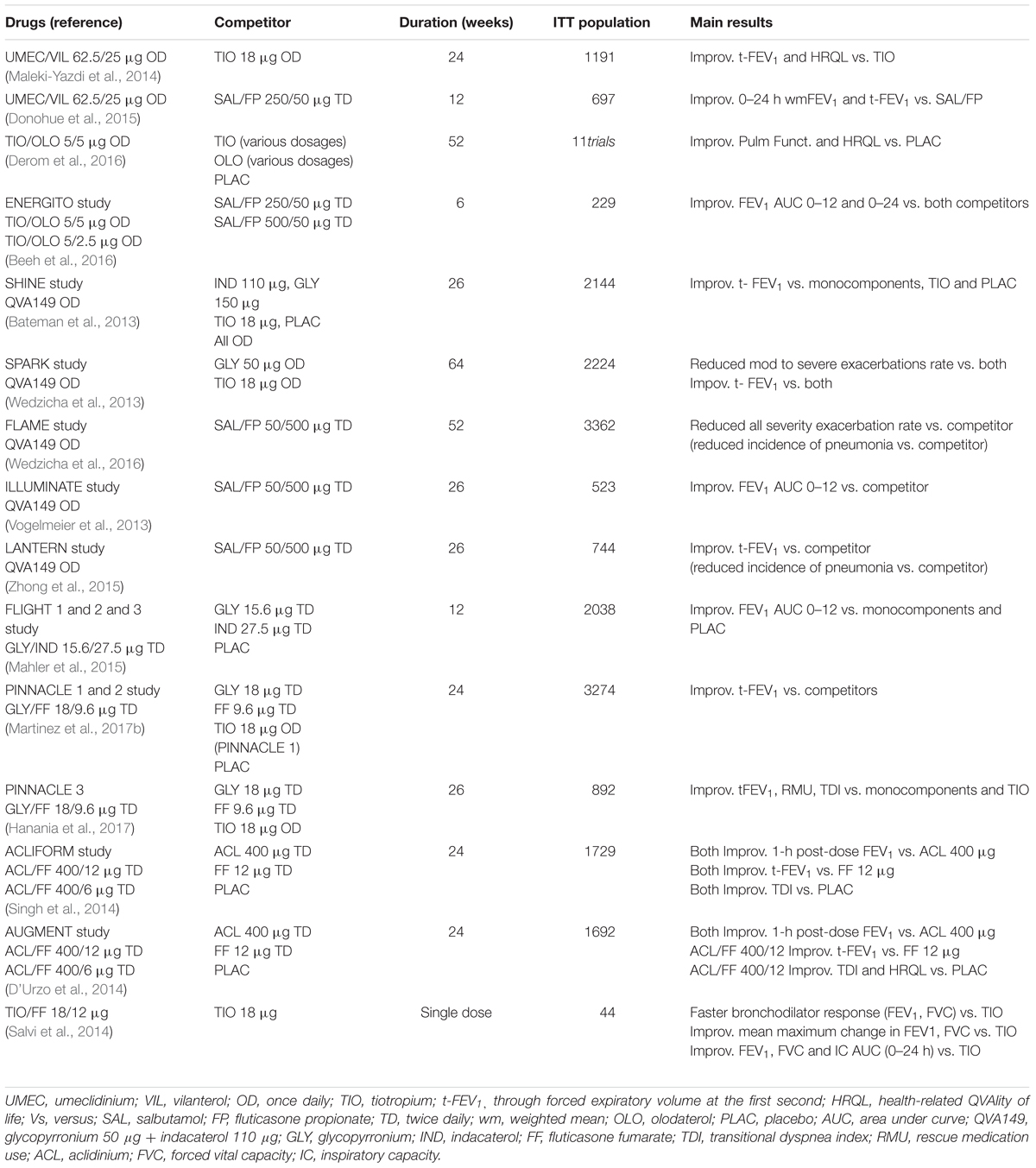
Triple ICS/LABA/LAMA FDC and dual LABA/LAMA or ICS/LABA FDCs are characterized by specific efficacy/safety profiles in agreement with the level of blood eosinophil count at baseline.ĬOPD ICS LABA LAMA dual bronchodilation network meta-analysis triple therapy. The weighted efficacy/safety profile resulting from SUCRA provided the following ranking in patients with low eosinophil count: ICS/LABA/LAMA>LABA/LAMA≫ICS/LABA whereas in patients with high eosinophil count the ranking was as follows: ICS/LABA/LAMA>LABA/LAMA>ICS/LABA FDC. COPD TRELEGY ELLIPTA 100/62.5/25 mcg is indicated in adult patients who are not adequately treated by a combination of an ICS/LABA or a combination of a LAMA/LABA: for the long-term, once daily, maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphys. 
The treatment ranking was reported via the surface under the cumulative ranking curve analysis (SUCRA).ĭata obtained from 21,909 COPD patients were extracted from the ETHOS, KRONOS, IMPACT, and TRILOGY studies, the only that fulfilled the strict inclusion criteria of this research. either dual LABA/LAMA or ICS/LABA therapies administered at fixed-dose combination (FDC) via the same inhaler device.Ī network meta-analysis was performed to assess the efficacy/safety impact of triple ICS/LABA/LAMA FDC compared with dual LABA/LAMA and ICS/LBA FDCs administered via the same inhaler device in COPD patients. The aim of this study was a quantitative synthesis by considering the studies that directly compared triple ICS/LABA/LAMA vs. FDA-regulated Device Product:ĭevice Product Not Approved or Cleared by U.S.In some studies comparing triple with dual combination therapies in COPD there might be a possible effect of inhaler bias resulting from different inhaler devices being used in comparator arms. studies in products where Boehringer Ingelheim is not the license holder studies regarding pharmaceutical formulations and associated analytical methods, and studies pertinent to pharmacokinetics using human biomaterials studies conducted in a single center or targeting rare diseases (in case of low number of patients and therefore limitations with anonymization). Individual Participant Data (IPD) Sharing Statement:Ĭlinical studies sponsored by Boehringer Ingelheim, phases I to IV, interventional and non-interventional, are in scope for sharing of the raw clinical study data and clinical study documents. Related Info Layout table for additonal information Pharmacy claims for non-index COPD maintenance medications on the index date.Pharmacy claims for multiple index medications on the index date.To restrict the cohort to first line maintenance therapy of Stiolto Respimat or first line maintenance therapy of Trelegy Ellipta we will exclude: patients on long-acting muscarinic antagonists (LAMA) monotherapy long-acting beta2 agonists (LABA) monotherapy inhaled corticosteroids (ICS) monotherapy free or FDC of: ICS+LABA, LAMA+LABA, ICS+LABA+LAMA therapy within six months prior to index date.To increase the likelihood of a true diagnosis of COPD, we will exclude all patients with two medical claims of asthma, cystic fibrosis, lung cancer, or interstitial lung disease in any position on separate dates of service during the study period.At least one year of continuous medical and pharmacy health plan eligibility prior to the index date is required (to allow a baseline period for the covariates and characterizing the study population).Two medical claims (at least one claim on index date or before in the baseline period) with an International Statistical Classification of Diseases and Related Health Problems (ICD)-9/10 diagnosis code(s) for chronic obstructive pulmonary disease (COPD) in any position during the study period (baseline ± post index date).For Trelegy Ellipta users, the first pharmacy claim of FDC of Fluticasone Furoate + Umeclidinium + Vilanterol (100/62.5/25 mcg), will be defined as the index date.For Stiolto Respimat users, the first pharmacy claim of Fixed Dose Combination (FDC) of Tiotropium + Olodaterol (5/5 micrograms (mcg)) will be defined as the index date. 
≥40 years of age as of the year of the index dateĪt least one pharmacy claim for Stiolto Respimat or Trelegy Ellipta.Why Should I Register and Submit Results?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
